The development of new drug molecules has never been easy and is full of challenges, some of which include the following:
- The time and cost of Research and Development (R&D) involved is high.
- Success of new drugs, like market acceptance and market capture, is quite uncertain.
- There are Government policies to take care of patients interest thereby regulating drug prices and allowing compulsory licensing.
- Challenges and intense competition from manufacturers of low-cost generics and me-too drugs of same therapeutic class.
Pharmaceutical industry is one of the most intensively regulated sectors and as a result of these challenges, returns on R&D investments in the pharmaceutical industry have decreased. One feasible alternative to address these challenges is to adopt strategies to reduce development costs and maximize profit. One such strategy is Product Differentiation. Product Differentiation is innovating an existing product, i.e., creating a product with better features, performance, or efficacy. Most blockbuster drugs have gone off-patent, and more are expected to go off-patent in the near future. Despite increasing R&D investments, new drugs to replace those that have gone off-patent or will sooner go off-patent are few in number. As a result, both innovator companies and generic drug makers need to focus on developing differentiated products to sustain growth in a challenging pharmaceutical sector, which otherwise sees price erosion, significantly increasing competition, and stringent regulations across the globe. Differentiated products paves the way for strengthening Product Pipeline and Life Cycle Management. The first step to distinguish one’s product from innovators and competitors is to think in all possible directions and innovate.

- Opportunity Identification
The adage that“better is always worth fighting for” could not hold more true than for the pharmaceutical business. Improving existing drugs, i.e., making them more effective, more patient-compliant, reducing dose and side-effects, is always welcome in the pharmaceutical and healthcare industry. New dosage forms, fixed drug combinations, new route, new strength, and new indications are the most exploited differentiation strategies. A good rationale supporting the development of a differentiated product is absolutely necessary, and rationale may even be a long felt need of improvisation. - Landscape and White Space Analysis
A thorough literature search and its review gives a fair idea of existing knowledge, technological know-how, and possible gaps. Landscaping includes both Patent and Non-Patent searches. This also helps in successful patent fencing of the product at later stages. - Design and Development
Product development is a set of activities beginning with the identification of a market opportunity and ending in the large-scale production and commercialization of a product. Depending on the targeted results and market of interest, one may follow the Bio availability-Bio equivalence route (BA/BE route) or the Clinical trial route. The United States Food and Drug Administration (U.S. FDA) provides for Section 505(b)(2) Regulatory Pathway filings for differentiated products. Section 505(b)(2) helps avoid unnecessary duplication of studies already performed on a previously approved (“reference” or “listed”) drug. Under this section, FDA can rely on data not developed by the New Drug Application (NDA) applicant. In simpler words, NDA applicant need not again establish the safety and efficacy of the chemical entity. This is a less expensive and much faster route to approval, compared with the traditional development path under Section 505(b)(1), which helps create new, differentiated products with significant commercial value. To enter the Indian market, one may have to simply conduct a bridging clinical trial (Phase 3) that saves time and is cost effective.
Depending on the targeted results and market of interest, one may follow the Bio availability-Bio equivalence route (BA/BE route) or the Clinical trial route. The United States Food and Drug Administration (U.S. FDA) provides for Section 505(b)(2) Regulatory Pathway filings for differentiated products. Section 505(b)(2) helps avoid unnecessary duplication of studies already performed on a previously approved (“reference” or “listed”) drug. Under this section, FDA can rely on data not developed by the New Drug Application (NDA) applicant. In simpler words, NDA applicant need not again establish the safety and efficacy of the chemical entity. This is a less expensive and much faster route to approval, compared with the traditional development path under Section 505(b)(1), which helps create new, differentiated products with significant commercial value. To enter the Indian market, one may have to simply conduct a bridging clinical trial (Phase 3) that saves time and is cost effective. - IP Protection
- Patenting – Patents encourage innovation and protect the interests of inventors. However, patenting differentiated products can be quite tricky, particularly, in developing countries like India that have a higher threshold for incremental innovations. Incremental inventions with a mere purpose to extend patent life and market monopoly rather than providing a solution to an existing need or problem are likely to be unpatentable. Patenting or Patent fencing of a differentiated product in comparison to patenting of a radical invention requires strategic thinking and excellent patent drafting skills.
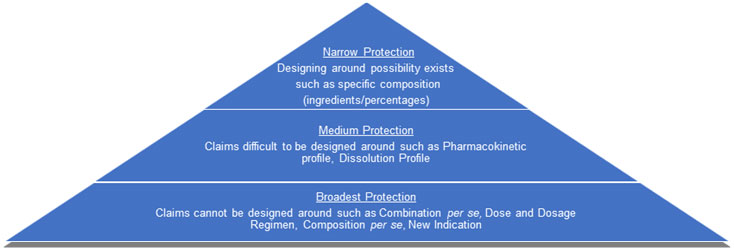
Patent Fencing Pyramid
It is rare in the case of differentiated products for a single patent to be so revolutionary so as to give you an edge over your competitors. On the one hand, broader claims may be difficult to obtain, enforce, and always have higher possibility of getting revoked. On the other hand, narrow claims may not be easy to revoke but could be designed around. A practical strategy is to use a mix of broad and narrow patents that limit competition, provide good enforcement, and may even provide out-licensing opportunities. Patent strategy becomes more complex and more difficult for competitors to crack as the number of patents in the portfolio increases. One patent rarely provides the same power and flexibility that holding multiple patents provide. Protect smartly keeping in mind maximum enforceability and minimum possibility for designing around.
Benefits of Patents:
- Patents on differentiated products can help better survive the market in form of market monopoly and limiting competition.
- Strategic patent filings could even form a part of the overall branding of the product.
- It justifies differential pricing of products as compared to their generic equivalents.
- The terms “Patent pending” or “Patented” convey that the products are unique and significant R&D efforts had been involved in its creation.
- Royalties and Out-licensing opportunities
Landscaping at early stages can also help an IP strategist to decide on offensive patent filings or defensive patent filings.
IP strategists pursue offensive patent filings to block competitors from using proprietary technologies as well as to generate royalties by enforcement of patent rights. Thus, it is advisable to file as many patents as possible covering all reasonable aspects. Offensive patent filings are mainly done where less is known in the art. Defensive patent filings are done to ensure that the inventors can use their own innovations and are not blocked by competitors patenting that technology, thereby, restricting them to use their own inventions. Defensive patent filings are preferred when literature is abundant and where modifications or improvements may not appear to be significant enough to withstand the patentability criteria and succeed in a patent grant. To avert future risks from competitors to stop you from pursuing your own invention by finding a technical advancement therein and filing a patent application on the same technology, IP strategists may recommend defensive patent filings. Many jurisdictions including China, Japan, Brazil, Australia provide utility models or petty patents for incremental innovations.Presently, India does not have any provisions for grant of utility model patents.
- Exclusivities – Going through the clinical route may also fetch market exclusivity such as New Indication Exclusivity, New Dosage Form Exclusivity, New Combination Exclusivity in countries like US.US provides 5 years (4+1) of NCE exclusivity along with many others such as ODE, GAIN etc. Europe provides up to 11 years (8+2+1) of exclusivity for New drugs. India presently does not provide any form of exclusivities for either pharmaceuticals or agro chemicals.There had been serious debates in India in last decade regarding the feasibility of introducing data exclusivities for these sectors. No stand has been taken so far.
- Patenting – Patents encourage innovation and protect the interests of inventors. However, patenting differentiated products can be quite tricky, particularly, in developing countries like India that have a higher threshold for incremental innovations. Incremental inventions with a mere purpose to extend patent life and market monopoly rather than providing a solution to an existing need or problem are likely to be unpatentable. Patenting or Patent fencing of a differentiated product in comparison to patenting of a radical invention requires strategic thinking and excellent patent drafting skills.
















